Blog
- Google+
Study into the effectiveness of Soda Ash and Litharge as fire assay fluxes for silica
Study into the effectiveness of Soda Ash and Litharge as fire assay fluxes for silica
By Kevin Renton, Assayer from the Western Australian School of Mines, 1964
Foreword
From time to time we hear comments about fire assaying that we find interesting. Rarely do we hear anything that we find enlightening. Often we hear comments that simply demonstrate a profound lack of understanding by the person making the comments; sometimes we hear a “revelation” about a fact that has been quite normal for us. Regardless of what the comments are, or whether we agree with them or not, it is a good that people have opinions and are trying to understand the fundamentals of the Fire Assay processes.
It is always interesting to hear the comments, not so much for the technical revelations, but to hear how the rest of the industry is performing their assays. Often there is no absolute right or wrong, and variations can be accommodated by the fire assay process and still provide useful results. Sometimes, we suspect, good assays are achieved in spite of the understanding of the people doing the assays, rather than because of them.
One comment we heard recently was to do with the main fire assay fluxes, especially Soda Ash and Litharge.
The comment was that Soda Ash (Na2CO3) was not effective at fusing silica, and that Litharge (PbO) was the flux to use to attack silica.
It was an interesting comment – I certainly disagreed with the comment about Soda Ash: I have the fortunate double of being a Chemist as well as an Assayer. (I finished a Chemistry Associateship at the Western Australian Institute of Technology (now Curtin University) and worked as a Chemist at the Government Chemical Laboratories of Western Australia.)
As a junior chemist I had the embarrassing experience of doing a sodium carbonate fusion in a fused silica crucible. The wall thickness on the crucible was about 3mm – much larger than any sample particle size – and the sodium carbonate chewed through the base. Embarassing, but an interesting and useful experience that came with having a dual background.
So I knew one part of the statement wasn’t correct. I also knew that litharge did form various silicate compounds, so I was curious as to how effective it was at actually digesting silica.
Test parameters
For the study I chose to use assay silica flour and mix in a 1 to 1 mole ratio with each of the soda ash and litharge. For ease of handling 20g of silica was taken, equating to 74 g of litharge and 36g soda ash. (This also highlighted an interesting point : generally most comments amongst people discussing fluxes just refer to masses – it generally is not considered that it takes twice as much litharge (by mass) to have the same molecular mass as soda ash.)
However, it was anticipated that the silica flour, being very fine with a nominal particle size of 75microns (but is possibly even much finer), would
- Possibly react more effectively than some real samples – as much as it is generally a stated requirement of sample preparation that samples have 90% also passing 75 microns, it still allows for 10% to be over that size. Furthermore, it is always a possibility that some samples don’t achieve the 90% minus 75 micron objective, and those samples need to be accommodated if possible. Silica, as quartz, often hosts gold and it is important to see if larger particles will be fused with a particular flux. Quartz, after all, is very hard and often is the last component ground i.e. quite probably is a big percentage of the allowable 10% oversize.
and
- Be difficult to observe if it has actually been chemically attacked or simply dispersed. Hopefully that could be seen in the slag that would be produced, but it had to be considered.
For those reasons silica sand was also used for another pair of tests. If a flux could digest larger particles than would be expected in an assay sample it would indeed be a suitable flux for real samples.
The sand that was used for the test was readily available as the flush material in the preparation area. The size of it was around 1.5mm, being twenty times the nominal particle size of assay samples.
So, four test samples were run:
Sample 1 : 20g of silica flour and 74g litharge
Sample 2 : 20g of silica sand and 74g litharge
Sample 3 : 20g of silica flour and 36g soda ash
Sample 4 : 20g of silica sand and 36g soda ash
- All equating to 1/3d of a mole of each compound
Test conditions and results
The samples were run through as normal assay samples i.e. heated gently with a finishing temperature of 1080oC and for a normal fire assay time of forty minutes.
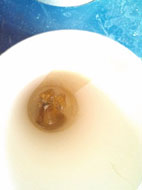
On first inspection the results seemed to favour the theory of the litharge being a good fusion flux provided the silica was very fine– only sample 1 appeared to have a suitable slag. See pics 1 to 4. However, the coarse silica clearly had not fused well. At this stage it was not possible to draw any conclusions about the soda ash fusions.
Pic 1 is the litharge and silica flour
Pic 2 is litharge and silica sand
Pic 3 is soda ash and silica flour
Pic 4 is soda ash and silica sand
However, a consideration of the relative melting points gave a possible answer to why the other samples did not fuse into good slags. Litharge has a melting point of 888oC, the lead silicate had a theoretical melting point of 766oC. If the lead and silica had fused it would have been quite fluid at 1080oC. Another possibility was that the litharge had simply melted and it could have had the silica flour simply dispersed through it. It was taken at face value that the litharge had reacted and fused with the silica flour. Further work needs to be done to investigate this.
However, the silica sand had not fused well, and in fact had granules of the silica still apparent in the flux (see pic 2). This is still possibly consistent with litharge fusing the silica, that the reaction was quite slow, and the particle size simply needed more time. (Note however that the samples were left in the furnace for a normal fire assay fusion time.)
It was virtually impossible to see what had happened with the soda ash and both the silica flour and the silica sand – both had formed a solid mass that wouldn’t pour at 1080oC. Soda ash melts at 850oC so, if the analogy with the litharge melting and dispersing the silica flour had happened, it would have to be assumed the same could happen with the soda ash. The more logical conclusion would be that the soda ash had reacted to form sodium silicate; the anticipated product of a one to one combination would have a melting point of 1088oC, hence not being fluid at the test temperature.
The litharge and silica flour sample was not treated any further, but the other three were placed back in the furnace at 1120oC for another half an hour.
All three poured from the crucibles, but the litharge and silica sand was not a good fusion. The effect is best shown from the following photographs:
Pic 5 Pic 6
Pic 5 is the litharge and silica sand after 30 minutes at 1120oC . Clearly the sand has not been fully attacked (but has had some sort of reaction to break the grains up).
Pic 6 is the soda ash and the silica sand after 30 minutes at 1120oC. The resultant slag was virtually a piece of clear glass, with no evidence of any particulate material. The slag was so clear the lines and the striations of the printing underneath could be clearly seen.
Two photos of the crucibles after the sand fusions also tell a story:
Pic 7 Pic 8
Pic 7 is litharge and silica sand after 1120oC – The lumps up the wall are of unfused silica sand.
Pic 8 is soda ash and silica sand after 1120oC – There is no evidence of silica particles.
Conclusions
There is some doubt from the studies whether the litharge was effective at reacting with the silica when it was very fine.
There is no doubt that the litharge did not totally fuse the silica sand with a particle size of 1.5mm, and in fact could not reasonably be described as having fused for any practical or effective purposes. Note the temperatures of the final fusion were greater than those in a normal finishing temperature of 1080oC , and well above the melting point of litharge.
The soda ash did totally fuse the silica, even with a particle size of 1.5mm. At what temperature the fusing started is not known, but it is presumed it had started by 1080oC (otherwise the unfused silica would have simply been dispersed in a matrix of molten soda ash). The mixtures of soda ash and silica were almost certainly unpourable at 1080oC simply because of the high melting point of the reaction product.
Comments
Soda ash is clearly a flux eminently suitable for fusing silica. The high melting point of the sodium silicate is obviously mitigated in a normal fire assay fusion by the other minerals and chemicals present, including more soda ash, borax, and litharge; all having lower melting points.
We are not arguing for people to not use litharge if they believe it does fuse silica.
However, these tests confirm our long-held beliefs and will not cause us to change from the philosophy of using soda ash to effect fusion on silica and silicates.
Further studies
This study has focussed on the fusion of silica. Quartz (SiO2) is a well known host for gold and hence it is imperative to effect good fusion of any silica in the sample.
However, silicates constitute a large portion of many oxide zone minerals and would also need to be effectively fused to ensure good liberation of any gold. A future study could replicate this study but with silicate minerals.
Yet further studies could study the effect of the fluxes on, say, iron oxides (especially magnetite) and sulphides.
Previous Article
AAS vs Classical GoldComment On Study into the effectiveness of Soda Ash and Litharge as fire assay fluxes for silica
Thank you for some other informative web site. Where else may just I am getting that type of information written in such an ideal means? I’ve a project that I’m simply now operating on, and I’ve been on the glance out for such information.
Please keep sharing :https://www.chemocart.com/search.php?search=Soda+Ash&search_result=search
Also read my blog : http://blog.chemocart.com